|
DFMDA
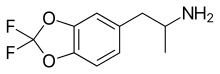
Difluoromethylenedioxyamphetamine (DFMDA, DiFMDA) is a substituted derivative of 3,4-methylenedioxyamphetamine (MDA), which was developed by Daniel Trachsel and coworkers, along with the corresponding fluorinated derivatives of MDMA, MDEA, BDB and MBDB, with the aim of finding a non-neurotoxic drug able to be used as a less harmful substitute for entactogenic drugs such as MDMA. Since a major route of the normal metabolism of these compounds is scission of the methylenedioxy ring, producing neurotoxic metabolites such as alpha-methyldopamine, it was hoped that the difluoromethylenedioxy bioisostere would show increased metabolic stability and less toxicity.[1][2][3] These compounds have not yet been tested in animals to verify whether they show similar pharmacological activity to the non-fluorinated parent compounds, although in vitro binding studies show DFMDA to have a SERT affinity in between that of MDA and MDMA.[4] It is also now generally accepted that MDMA neurotoxicity results from a variety of different causes and is not solely due to accumulation of alpha-methyldopamine,[5][6][7] making it unclear how much less neurotoxic DFMDA and related drugs would be in practice. References
|
||||||||||||||||||||||||||||||||||||||